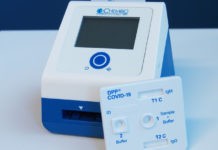
Following GenScript Biotech Corporation cPass SARS-CoV-2 neutralization antibody detection kit has received provisional authorization by Health Science Authority (HSA) Singapore on 8 May 2020, CE-IVD marking in Europe on 26 May 2020, and FDA approval in the Philippines for commercial use on 11 August 2020.
US Food and Drug Administration (FDA) issued an emergency use authorization (EUA) for cPass SARS-CoV-2 Neutralization Antibody Detection Kit on 6 November 2020.
cPass SARS-CoV-2 Neutralization Antibody Detection Kit, which is co-developed with Duke-NUS Medical School Singapore and Diagnostics Development Hub from Singapore’s Agency for Science, Technology, and Research (A*STAR) and manufactured by GenScript Biotech Corporation, is said to be the first serology test authorized to detect neutralizing antibodies – the specific antibodies present in the serum of Covid-19 patients that are responsible for clearing the viral infection, without the need of live biological materials and biocontainment facility. The test measures the presence of neutralizing antibodies in patients recovering from Covid-19 or receiving a vaccine.
GenScript has started to serve the global market through authorized distributors and authorized laboratories to test the public. GenScript promised to support the healthcare community and continue the fight against Covid-19.
GenScript Biotech Corporation is a global biotechnology group. Based on its leading gene synthesis technology, GenScript has developed four major platforms, including the global cell therapy platform, the biologics contract development and manufacturing organization (CDMO) platform, the contract research organization (CRO) platform, and the industrial synthesis product platform.
GenScript was founded in New Jersey, US, in 2002 and listed on the Hong Kong Stock Exchange in 2015. GenScript’s business operation spans over 100 countries and regions worldwide, with legal entities located in the US, Mainland China, Hong Kong, Japan, Singapore, Netherlands, and Ireland. GenScript has provided premium, convenient, and reliable products, and services for over 100,000 customers.
GenScript has several intellectual property rights and technical secrets, including more than 100 patents and over 270 patent applications. As of June 30, 2020, GenScript’s products and services have been cited by 51,000 peer-reviewed journal articles worldwide.