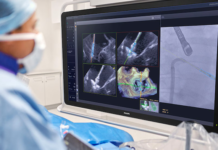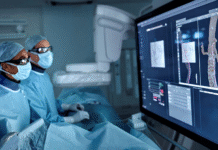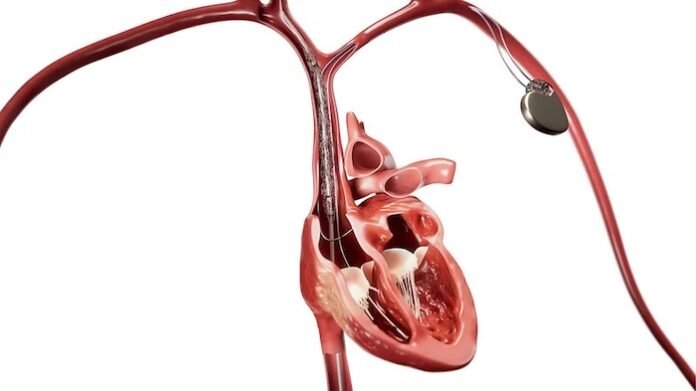
Royal Philips, a global leader in health technology, announced the launch of the next-generation Bridge Plus Occlusion Balloon, designed to help electrophysiology teams rapidly control bleeding and stabilize patients during rare but life-threatening superior vena cava (SVC) tears in transvenous lead extraction (TLE) procedures. Building on technology used in more than 50,000 procedures, Bridge Plus enables electrophysiology teams to respond in minutes – helping stabilize patients and buy critical time for surgical repair.
Lead extraction procedures are commonly performed to remove leads from cardiac implantable electronic devices (CIEDs), such as pacemakers or defibrillators, due to damaged, infected or malfunctioning leads. While there is evidence of lead extraction’s safe use, SVC tears, occurring in fewer than 0.5% of cases, can quickly become fatal without immediate intervention.
Bridge Plus is designed to provide temporary vessel occlusion, helping reduce blood loss, maintain hemostasis, and stabilize patients during emergencies. The balloon can deploy in less than two minutes, stop up to 90% blood loss, and maintain acceptable hemostasis for at least 30 minutes. This provides physicians with critical time to stabilize patients and prepare for surgical repair.
“Ensuring procedural safety is a top priority for electrophysiology teams,” said Stacy Beske, business leader, image-guided therapy devices at Philips. “Bridge Plus builds on established technology to help physicians prepare for rare SVC tears and respond quickly with the control needed to stabilize patients and transition to surgery.”
Clinical evidence highlights the importance of being prepared for this emergency scenario, with studies showing that survival rates in SVC tear events improved from 56.9% to 88.2% when an occlusion balloon was staged and available during procedures
“Although superior vena cava tears are rare, they represent one of the most critical emergencies that can occur during lead extraction,” said Thomas Callahan, an electrophysiologist at Cleveland Clinic who studied the technology. “Having an occlusion balloon staged and ready can significantly improve response time when every second matters. This technology may help teams prepare for these rare but serious events and support safer lead extraction procedures.”
Prophylactic balloon set up brings additional benefits during TLE procedures, including individualized balloon staging for each patient, and the ability to deploy Bridge Plus if fluoroscopy is no longer available.
Bridge Plus is a low-pressure, compliant occlusion balloon with radiopaque markers designed for accurate placement and compatibility with a wide range of patient anatomies [9]. The device expands Philips’ portfolio of lead management solutions supporting safe and effective extraction procedures.
The Bridge Plus Occlusion Balloon is now commercially available in the United States, with international availability expected later in 2026, pending country registrations. The solution will also be showcased at Heart Rhythm Society (HRS) 2026, taking place April 24-26 in Chicago, IL.